Boffins snap X-ray closeup of single atom – and by closeup we mean nanometres
Achievement took 12 years of blood, sweat and science
Scientists in the US have managed to capture the first X-ray image of a single atom, and it only took 12 years of work developing a technique and a super-powered X-ray instrument to do it.
Now, after a dozen years of work, the team of scientists from Ohio University, Argonne National Laboratory, the University of Illinois-Chicago and other institutions have captured an X-ray image of an actual iron atom that, unlike previous atomic-scale snapshots, actually allows for the individual atom to be identified.
"Atoms can be routinely imaged with scanning probe microscopes, but without X-rays one cannot tell what they are made of. We can now detect exactly the type of a particular atom, one atom at a time, and can simultaneously measure its chemical state," said Saw Wai Hla, an Argonne scientist, Ohio U professor of physics and head of the Nanoscale and Quantum Phenomena Institute at OU, as well as the lead scientist on the project.
Fresh technique
The microscope purpose-built for this new type of imaging uses a technique known as synchrotron X-ray scanning tunneling microscopy, or SX-STM. That technique is itself a combination of scanning probes traditionally used to take pictures of amorphous atoms, and synchrotron X-ray imaging, which uses a far more brilliant form of radiation that's able to be controlled well enough to snap X-ray images at the atomic level.
Scanning tunneling microscopy can take fantastically microscopic images by bringing the tip of an instrument incredibly close to the surface of an object – mere nanometers away – and using an electric current to agitate the atoms in its target. While great at generating bumps and nanoscopic waveforms, there's no way to get an actual identification of the atoms being scanned.
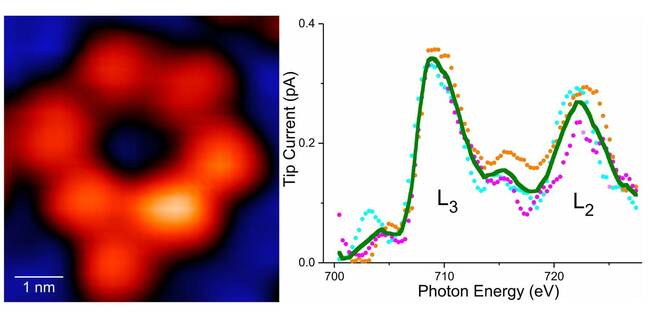
A X-ray image (left) of a supramolecule containing one atom of iron and associated X-ray signature (right) - Click to enlarge
Enter synchrotron radiation, which can be powerful enough to penetrate deep into matter and study features like molecular bonds or individual atoms.
- Brits and Yanks join forces to make fusion magnets cool again
- CERN spots Higgs boson decay breaking the rules
- LIGO cranks up the sensitivity to sniff out gravitational waves
- Encoded 'alien message' will reach Earth today, but relax: It's just a drill
Combine your scanning tunnel microscope and synchrotron radiation, and you're able to image atoms via "photoabsorption of core level electrons," which the researchers said means they can capture the elemental fingerprints of an individual atom. In this case, an iron atom was used for the experiment.
"Using X-rays to detect and characterize individual atoms could revolutionize research and give birth to new technologies in areas such as quantum information and the detection of trace elements in environmental and medical research," said Ohio University PhD candidate Tolulope Michael Ajayi, the lead author on the study.
A rare-earth accomplishment
In order to capture the image, the team had to create a supramolecule made of a single iron atom and several terbium atoms. Along with capturing the image of iron, Hla's team was also on the lookout for how different environments affect single rare-earth atoms, like terbium.
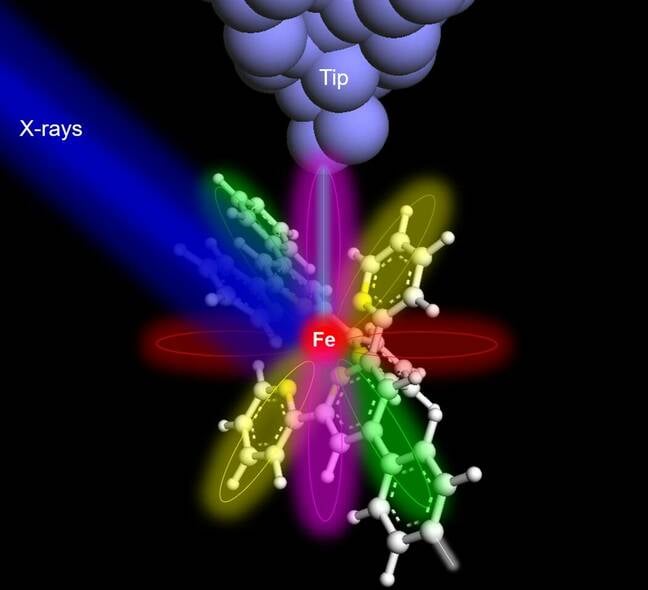
A diagram displaying the X-ray of the supramolecule: X-rays are shot at the molecule, and electron agitation causes tunneling effects detected by the tip to identify the specific molecule
"By comparing the chemical states of an iron atom and a terbium atom inside respective molecular hosts, we find that the terbium atom, a rare-earth metal, is rather isolated and does not change its chemical state while the iron atom strongly interacts with its surrounding," Hla said.
That means the team has essentially figured out how to not only identify individual elements at the atomic level, but their chemical states as well. They've also discovered a way to apply their technology to a new technique called "X-ray excited resonance tunneling," which they say makes it possible to detect the orbital orientation of a single molecule on a material's surface.
Hla said that discovery could allow future researchers to experiment with the "quantum and spin (magnetic) properties of just one atom using synchrotron X-rays." Going forward, the team plans to look into additional ways to apply the technology to areas like researching the gathering of critical materials and other atomic advancements. ®
